H3BO3 Lewis Structure: How to Draw the Lewis Structure for B(OH)3

H3BO3 Lewis Structure: How to Draw the Lewis Structure for B(OH)3
A step-by-step explanation of how to draw the B(OH)3 Lewis Dot Structure (Boric Acid, also called H3BO3).Note that Boron (B) is an exception to the octet rul

⏩SOLVED:Draw the Lewis structure of boric acid, B(OH)3. (a) Is…

SOLVED: Boric acid, H3BO3, also written as B(OH)3, acts as a weak acid in water. However, it does not do so by loss of a hydrogen ion. Instead, it acts as a

Explain the nature of boric acid as a Lewis acid in water.
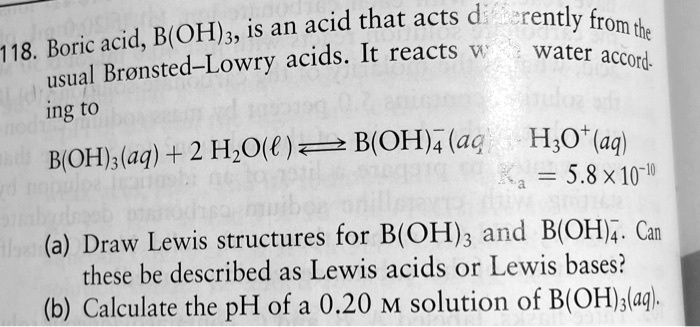
SOLVED: Boric acid is an acid that acts differently from the acid B(OH)3, the 118th Boric Bronsted-Lowry acid. It reacts with water according to the usual equation: B(OH)3(aq) + H2O(l) â†' B(OH)4-(aq) +

Boric acid molecule formula hand drawn imitation Vector Image

Boric acid is weak monobasic acid and acts as Lewis acid

SOLUTION: Ass5 memo - Studypool

Solved Help needed for Organic Chemistry . Please try to
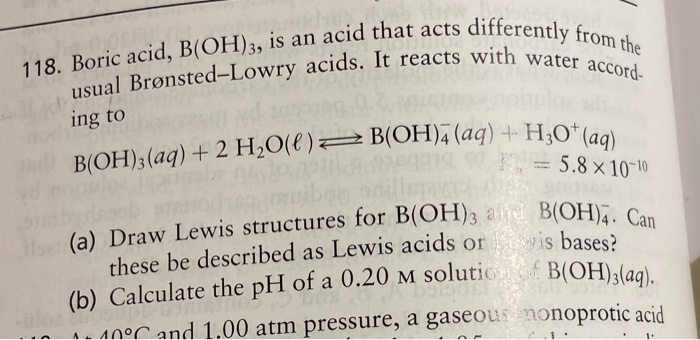
Solved 77. Thiamine hydrochloride (vitamin B, hydrochloride

How to determine the Lewis dot structure of H3BO3 - Quora

Solved 3) Using Lewis structures and curved arrows, write


